" Chem: what is it GOOD for?"
Absolutely EVERYthing (say it again, y'all). --with thanks to Edwin Starr
If you have ever taught, especially at the undergrad or pre-college level, especially in courses like science or math, you always have at least one kid you says: “Why do we need to learn this? When am I ever going to use this?” Those of us who are veteran teachers have a variety of responses, like the classic “it’s the intellectual version of going to gym and lifting weights” or some other version of “shut up, kid”. In the late 1970s, I was working in military medicine and tutoring student nurses in chemistry and math on the side. Those poor (mostly) girls did not the luxury to spend even a single second thinking about why they were doing organic chemistry or learning stoichiometry. They were desperately trying to survive, just enough to pass to be able to move through their program. That’s certainly the case for many academic majors — you really do not have the time to reflect on what you are learning and why you are learning it. That’s a shame, but it’s the way the system works. (AI is making this worse, but that’s a different discussion.) You have probably taken chemistry at some point in your life, but it’s likely that there was never any effort made on anybody’s part to help you understand why what you were learning was important, and important in every day life. You just wanted to get through it.
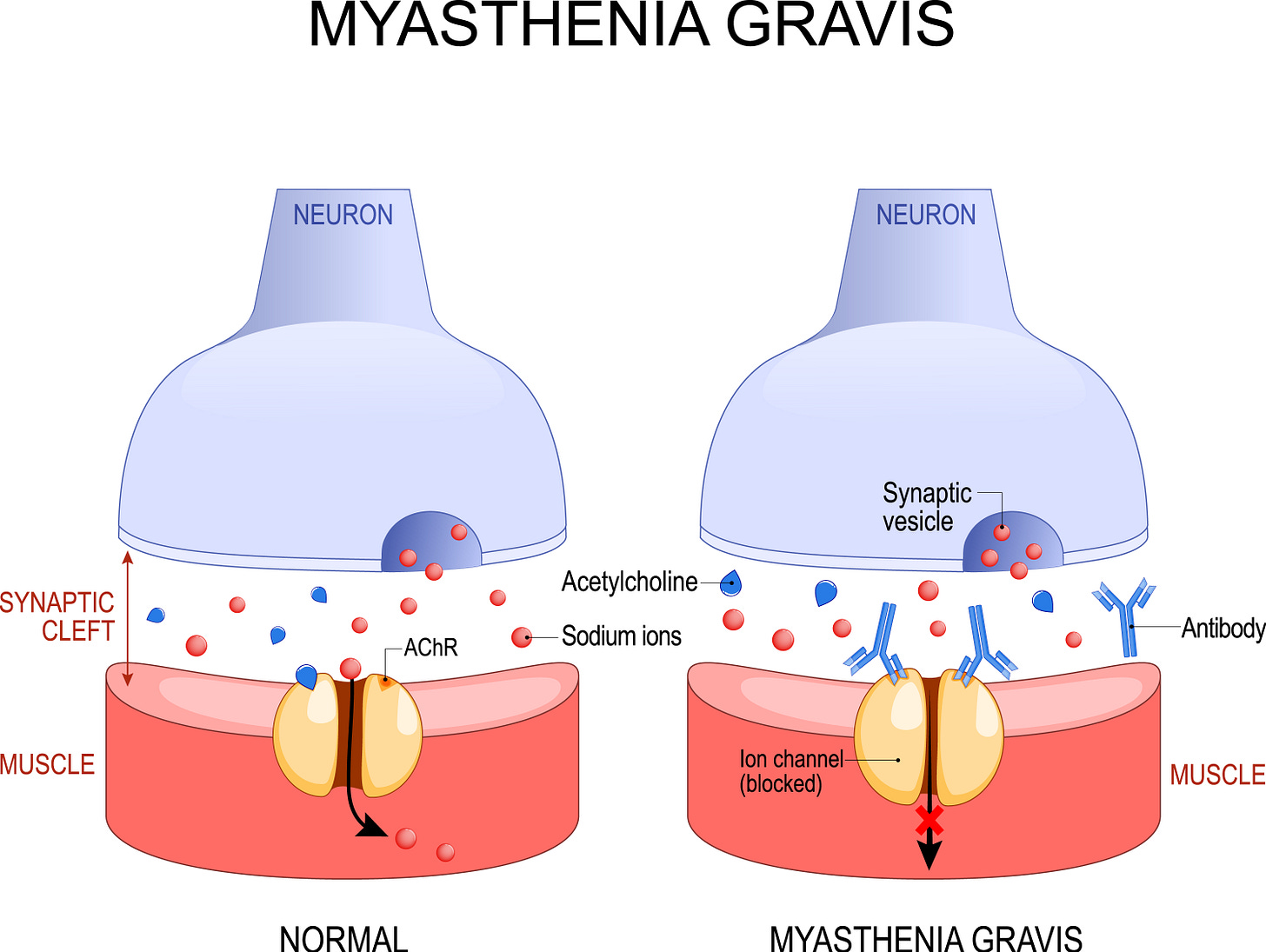
As I’m writing this, I’m sitting in a lovely hospital bed on the 4th floor of Durham Regional Hospital (an affiliate of Duke Medical Center, or “Little Duke” as it’s known in town). I’m here getting treated for issues related to my myasthenia gravis. MG is an autoimmune neuromuscular disorder, resulting in various muscular symptoms, such as eyelid drooping (dysplopia), difficult swallowing (dysarthria), and difficult speaking (dysphagia). My particular disorder is “ocularbulbar”, meaning affecting the muscles in the head and the neck, the most common type of MG, hence the symptoms in those areas.
The cause for this disorder is that my body’s own antibodies block a neurotransmitter called acetylcholine from binding to muscle fiber receptors, resulting in reduced muscular function. . The graphic below shows the release of acetylcholine (Ach) being released from a neuron and binding to the acetylcholine receptor (AchR) on the muscle that triggers contractions. In my case, my own antibodies are blocking access to those receptors, reducing the ability of my muscles to do what they are supposed to be doing. Bummer.
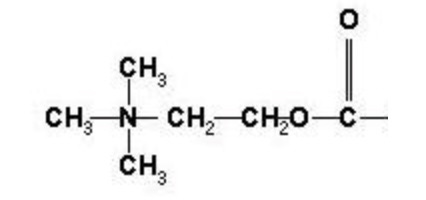
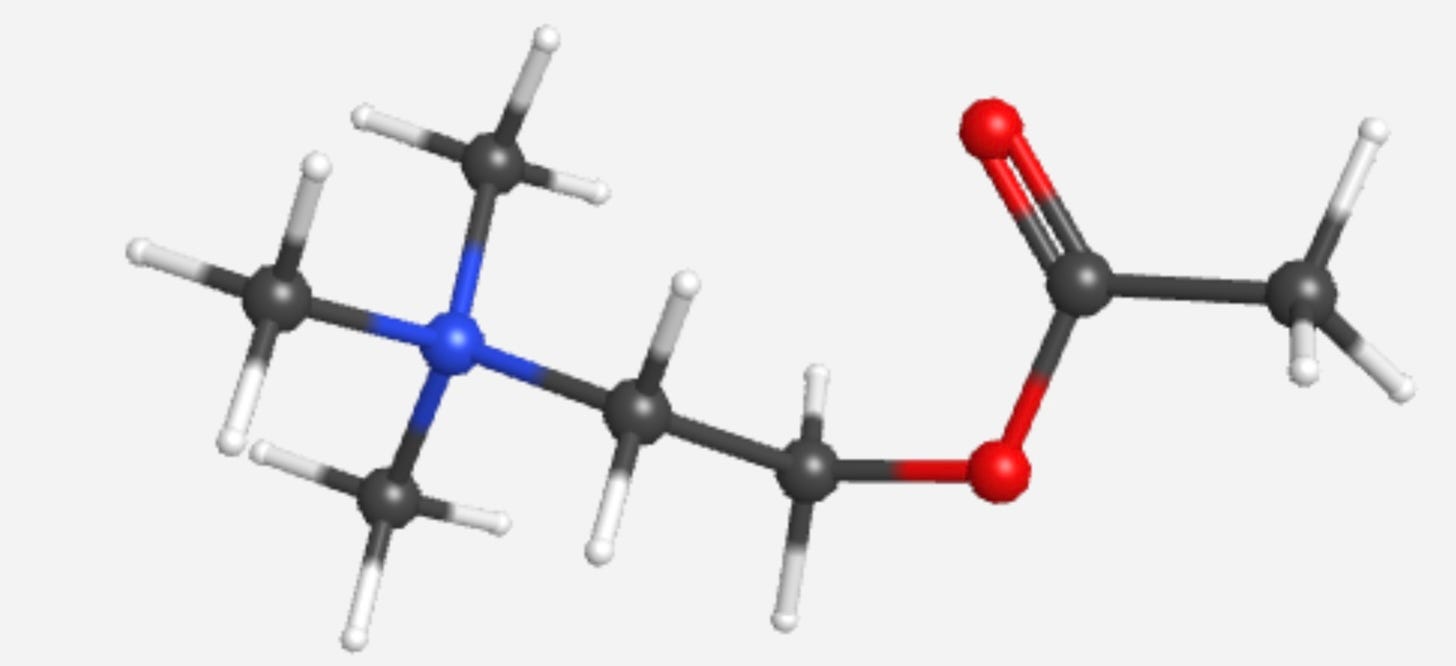
Acetylcholine is an endogenous molecule, meaning it’s not a drug we take — it’s already in our body. It’s an organic molecule, meaning it’s primarily carbon (C), but also with some nitrogen (N), oxygen (O), and hydrogen (H). I show two representations, one an expanded chemical formula showing the bonds, and one “ball and stick” model, where the carbons are gray, oxygens red, nitrogens blue, and hydrogens white.
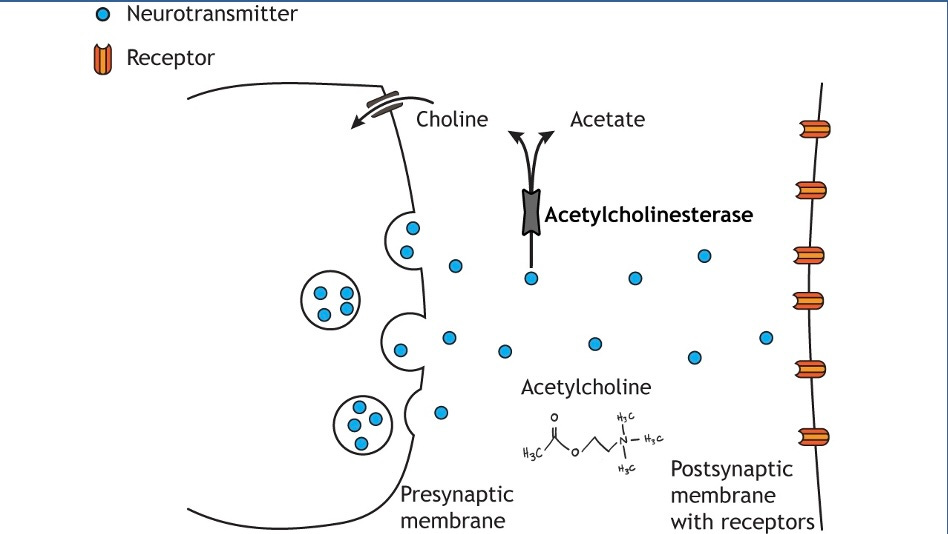
Because acetylcholine is a pretty important neurotransmitter for muscle function, and I have antibodies blocking the receptors, I need as much acetylcholine as I can get. Unfortunately, acetylcholine undergoes a chemical reaction in the brain that breaks it down to acetate and choline. The chemical that does this is a protein called “acetylcholinesterase”, an enzyme (I know this because the name ends in “-ase”).
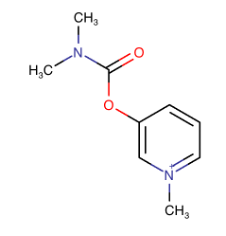
So, I take a drug called pyridiostigmine (“Mestinon”), shown below. This is an acetylcholinesterase inhibitor, and it prevents acetylcholine from breaking down into its two products. Yay for me, more acetylcholine. I notice within 30-45 minutes after taking this drug that things are working better. I can swallow, open my eyelids pretty much all the way, and talk to my wife and students with them being able to understand me. That’s a good thing.
But hey, most of you are healthy and pharmaceutical chemistry is not a central part of your daily life, like it is mine. Sure, you might pop a couple ibuprofen when you’ve been a little overzealous in the gym, but you don’t plan your life around medications. So, we’re going to use an example of how chemistry works in YOUR everyday life that does NOT include drug chemistry. That’s sort of the point of this post — chemistry in everyday life.
When I was thinking about this post, I probably had 18 or so examples of how chemistry is important to YOU in your everyday life; choosing one was a bit of a challenge. I looked at topics such as batteries, baking and cooking, microwave ovens, and several others. I finally settled, however, on a topic that is a little more interesting (at least to me): how self-darkening, or photochromic, glasses work. It’s really interesting chemistry, and lends itself to the use of computational science, which is, of course, the entire point of Confessions of a Computational Scientist. The other topics are OK, but not really targets for computational chemistry.
Many people who wear glasses don’t like to have to carry two pairs of glasses, one for inside, and one for outside in the sun. Me, I’m cheap, so I use clip-on sunglasses, which my wife hates, says they make me look like a dork. She’s not wrong.
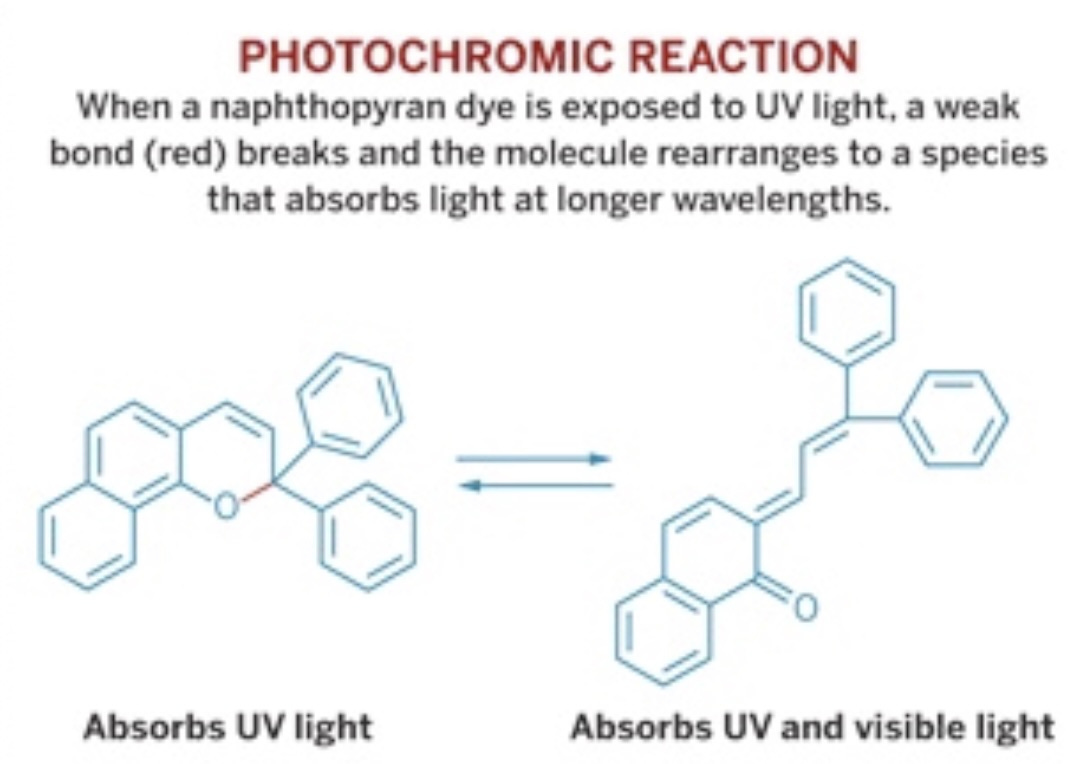
Self-darkening sunglasses, as the name suggests, are those glasses that change color (darken) in response to ultraviolet (UV) light, the light coming from the sun. Self-darkening glasses contain a photochromatic (photo: light; chromatic; color) chemical compound known as napthopyran. This compound is a relatively large organic (carbon-containing) compound that contains five rings with an oxygen atom embedded in one of the rings (see the graphic below). Upon exposure to ultraviolet light, one of the oxygen bonds breaks (the red-colored bond in the napthopyran, connecting the oxygen to the rest of the molecule), resulting in a lengthening of the structure. This new compound, indendonapthopyran, has different properties than the napthopyran. Specifically, napthopyran only absorbs radiation in the UV range of 200 nanometers (nm), while indendonapthopyran absorbs radiation in both the UV region and in the visible region (400-700 nm).
Notice in this reaction that the reaction arrows point both ways. This means napthopyran can turn into indendonapthopyran, and indendo can turn back into naptho. Cool.
It might be useful to back up a little and do a little science review. If you know this part, skip ahead!
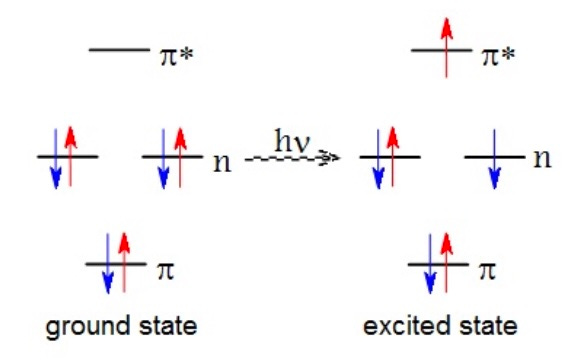
Atoms and molecules (combinations of atoms) contain electrons, very small negatively charged particles that are found near the nucleus of an atom, which is positively charged. Electrons live in “orbitals”, which represent energy levels. Each orbital can have a maximum of two electrons; think of an orbital as a two-seater sports car. When they occupy the same orbital, they must have opposite spin, a guy named Pauli said that. We represent the electrons with arrows, and the direction indicates spin.
Electrons want to be in the lowest energy state possible, sort of like the teenagers that I teach. On the lefthand side of this graphic, electrons are in the ground state. If I hit the electrons with light energy, represented by the “hv” notation, one (or more) of the electrons will “jump” up to a higher energy level, the excited state. Take away the light energy, and the electron(s) will return to the ground state. You don’t need to worry about the other notation stuff, such as “n” and and the “pi/pi*” symbol.
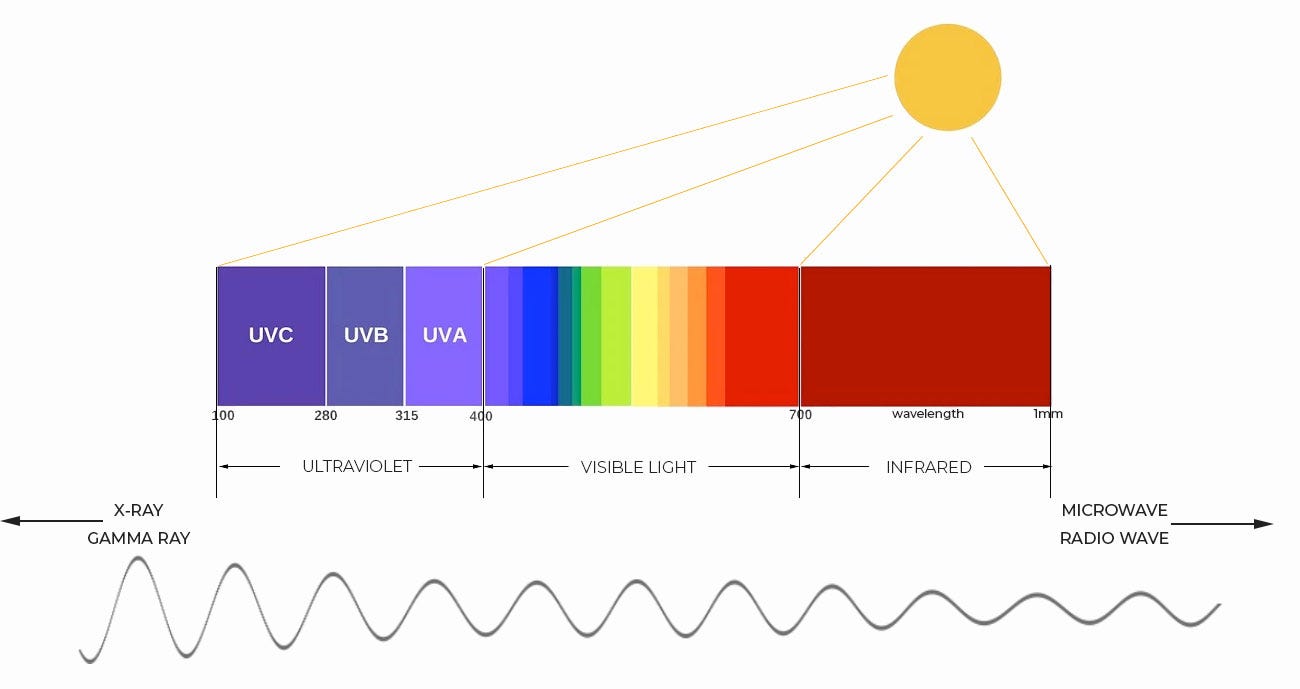
In our case, the light energy that is hitting the electrons comes from the sun, and has several types: ultraviolet (UV), visible, and infrared (IR). As we move from left to right on the UV-VIS-IR section of the electromagnetic spectrum shown below we have UV (UVC, UVB, and UVA), visible, and infrared. Going from left to right, the shorter wavelengths of UV radiation get longer, until they become long IR wavelengths. If we were to look at the entire spectrum, we’d see that even longer wavelengths are microwave and radio waves. Both useful in everyday like. I use both everyday in my microwave oven and in my car radio. So do you. Yay, chemistry (rats…I should say physics):
Notice that we have different types of light energy coming from the Sun: ultraviolet (UV), visible, and infrared (IR). We can only see the visible light, and going from right to left, we use the acronym ROYGBIV to remember the order: red orange yellow green blue indigo violet. IR radiation, which goes from 700 nanometers (nm) to about one (1) millimeter (mm) in length of the wavelength, can cause skin burns and damage to eyes.
For glasses, we are interested in the UV side of the spectrum, which goes right to left (longer to shorter) in wavelengths from 400 nm to 100 nm. There are three varieties of UV radiation: UVA, UVB, and UVC. Each corresponds to a part of the UV wavelength range, with UVA having the longest wavelengths and UVC the shortest.
Now it’s time to bring computational chemistry into play to study photochromic glasses (the paid subscribers will do these calculations directly; I’m showing you results here).
Using our computational chemistry server (http://chemistry.ncssm.edu), we build each of the molecules shown above: napthopyran and indendonapthopyran. Once done, we request that the comp chem server perform an “Excited States and UV-VIS” calculation using a piece of quantum chemisty software known as Gaussian16 (https://gaussian.com/). We use a technique called ZINDO: Zerner’s intermediate neglect of differential overlap, which, if you are a computational chemistry student, you will spend time learning about. Luckily for you, you’re probably NOT a computational chemistry student, so you don’t have to learn it. Phew!
We asked the server to calculate 40 different excited states. The more you do, the more accurate your results will be, but more calculations also requires more compute time. More would be necessary if you were doing research for publication, but it isn’t for a teaching unit.
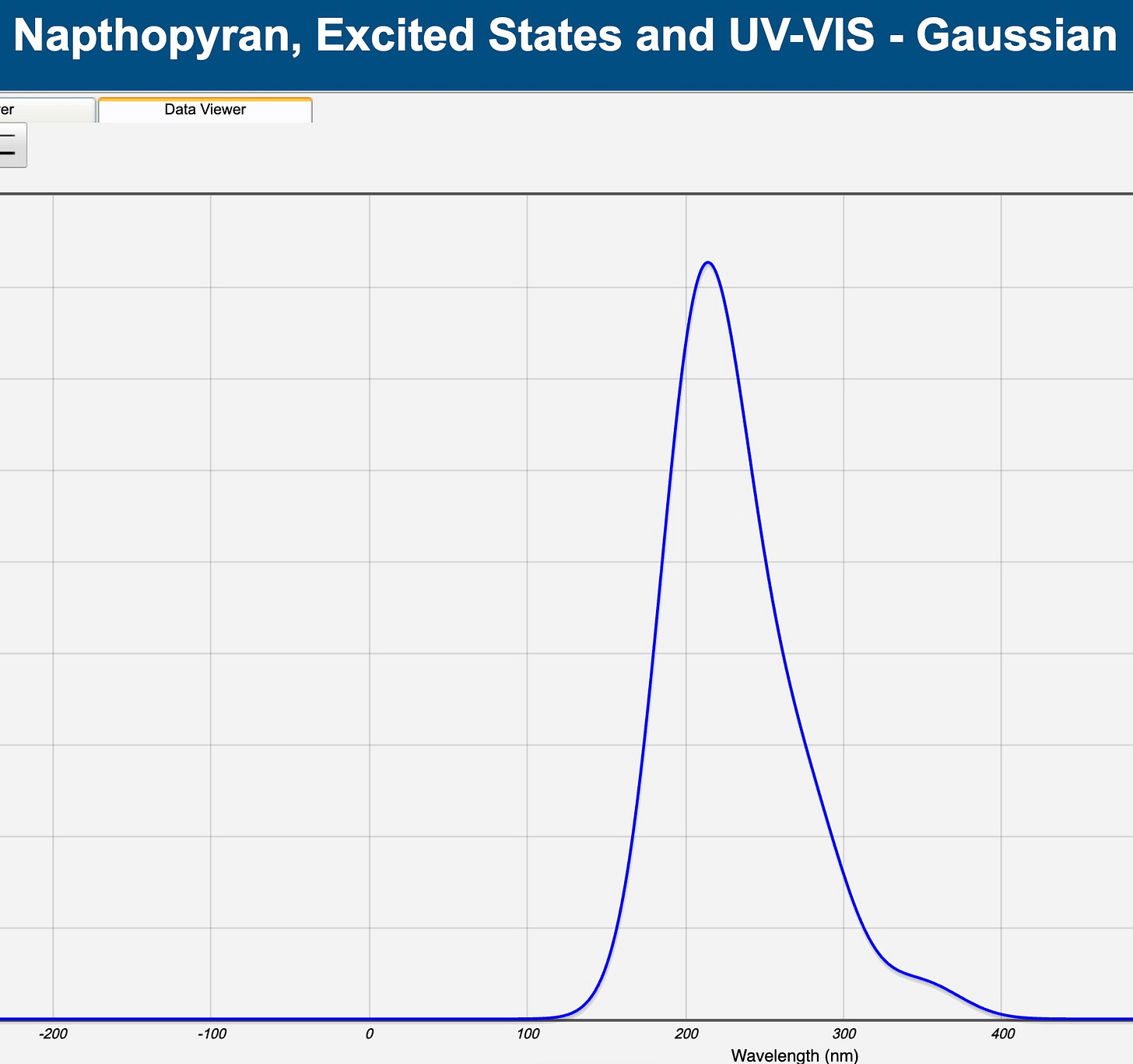
Here’s what the server told us: it said that most of the UV radiation was absorbed in Excited State 22 (out of 40). Lots of data here, and I won’t do all of it, but here’s the important stuff. The “f” number — 0.5047 — is the oscillator strength, a measure of the impact of the UV absorption. The other oscillator strengths were much lower, suggesting that they were not very important. The associated wavelength for this is 207.56 nm. Looking back at the UV-VIS-IR spectrum above, this puts us pretty squarely in the UVC range. In this range, the glasses are absorbing UV radiation at that wavelength, and our glasses are clear.
We can show these results as a graph, with the wavelengths on the x-axis, and the oscillator strength on the y-axis. There is clearly a big peak at a little over 200 nm, and I could zoom in to see the 207 nm explicitly.
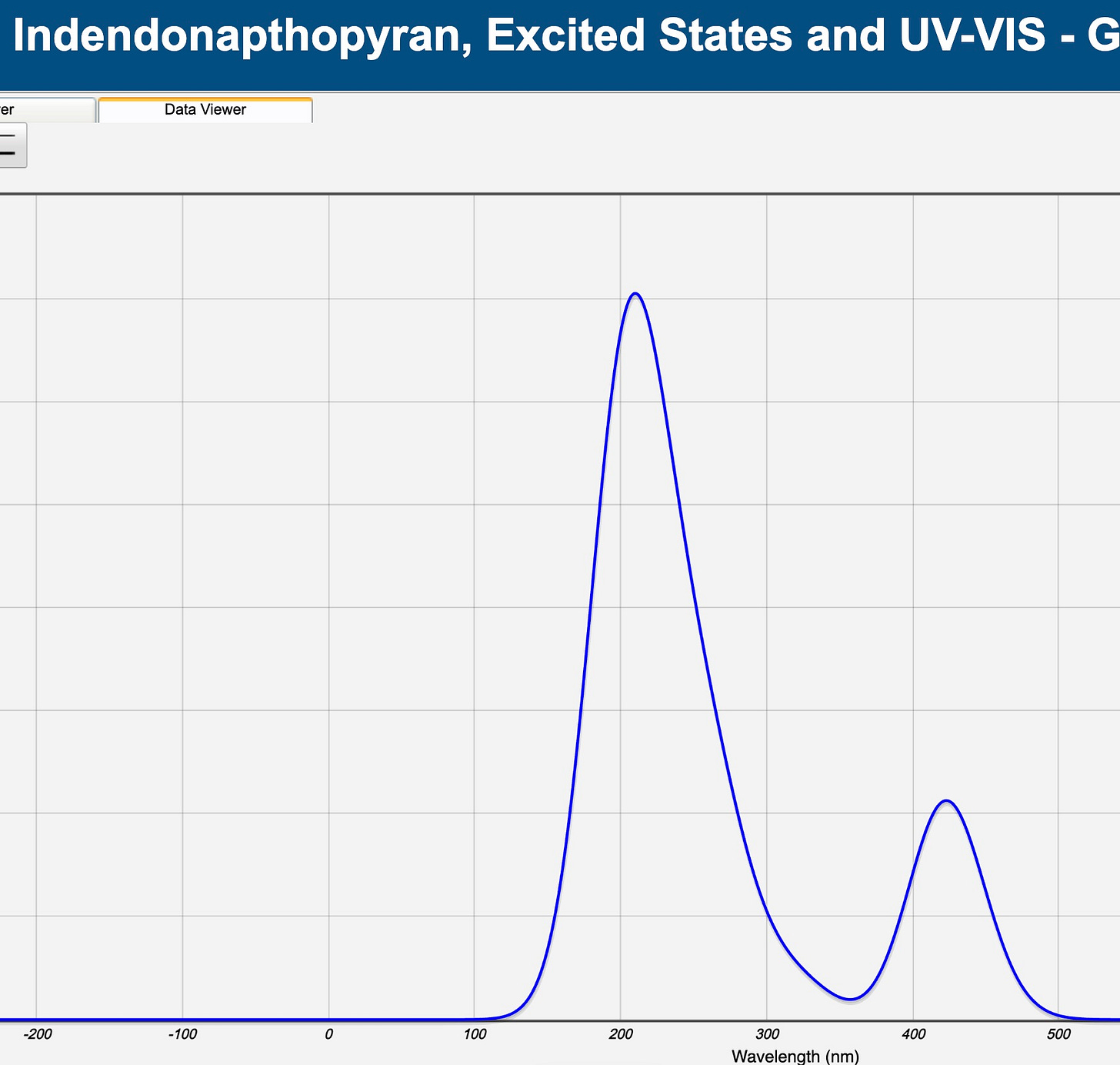
Now we do the same thing with the indendonapthopyran. Like with the napthopyran, we see a peak around 210 nm or so, but we also see a peak around 420 nm. Take a look at the UV-VIS-IR spectrum above. Where is 420 nm? It’s in the visible part of the spectrum. So the 210 nm is the napthopyran — the closed form of the compound, with the oxygen bridge intact — absorbing UV radiation, and the 420 nm is the indendonapthopyran —the open form of the compound with the oxygen bridge broken — absorbing the visible radiation. 420 nm is dark, violet-bluish, so the glasses appear dark. [NOTE! If you were to look up these numbers on Wikipedia or ChatGPT, they would be lower; the calculations from the comp chem server have some amount of error, so when we do a calculation using excited states/UV-Vis, we typically apply a “fudge factor” of about 11%, getting us to about 380 nm for the indendonapthopyran value that you would see in ChatGPT for experimental data.]
These kinds of computational studies allow us to study individual atoms, individual molecules, or, in this case, reaction mechanisms, such as the absorption of electromagnetic energy in the naptho-indendo reaction. There is a strong connection between what an experimental (bench) chemist does and what a computational chemist does. Computational chemists can run "theoretical” experiments, which can tell us that a chemical system or reaction can exist or happen. If our results suggest that the system/reaction won’t work, then we save the experimental chemist a great deal of time and expense. Getting back to the original conversation in this post, finding a new drug used to be egregiously time-consuming and slow. Now, we can use computational methods in drug design and discovery (a course I teach) to screen thousands and millions of potential drugs (a method known as high-throughput screening, or HTS). If our computational results suggest a “hit”, THEN we can go into the lab, synthesize that drug, and start testing it in Petri dishes, small animals, and, hopefully, eventually humans.
OK, thanks, I feel better. I’ve presented a number of different topics in Confessions, and it’s nice to “run home to Mama” every once in a while — after all, I am a computational/physical chemist. I love the “xkcd” cartoon series, and this cartoon on “purity” is pretty funny, but it’s also really true. What you saw in the UV study is just applied physics, specifically quantum physics. Us chemists like to say “chemistry is the central science”, then the physicists remind us the chemistry is just the “physics of electrons”, and then the math people stomp on all of us. Fortunately or unfortunately, the mathematicians win.
Next month we’re going to cross back over into the social sciences, and take a look at computational history. We’ll look at using computational tools to study the Civil War and also take a look at presidential elections from the past two or three cycles.
On the paid side, folks will use a real computational chemistry server to do a case study on the photochromic glasses. Join us! Until next time, have a great April, and don’t forget your taxes!